Processes in the Pharmaceutical Industry

Bulk Production
The bulk production of solid, semi-solid or liquid forms presumes a mapping of fixed batch sizes or of plants to be used alternatively and the resulting material flow. Depending on the modelling in the SAP system, it can be necessary to network several production steps with one another and clearly image the material flow (availability testing and ranges) on the process step level.
Campaigns often have to be created in order to link the production of the same products with each other and, if one order is moved, to move all other orders with it. ORSOFT supports you here with its campaign planning functionality, which makes it possible to create both horizontal campaigns (same product/same product group) and vertical campaigns (all products in a logistics chain).

Filling
In pharmaceutical production, an active ingredient, especially in liquid dosage forms, is divided into different volumes and bottling forms, which in turn are divided into different packaging sizes (and these into regional specifications).
In order to carry out production effectively, bottling and packaging forms are combined in planning into campaigns that are served by one approach. The aim is to reduce set-up costs or to meet regulatory requirements. These campaigns are often saved as collective orders in SAP PP-PI and are linked by external dependencies.
The combination of several process orders into campaigns is very complex and requires a great deal of expertise and experience. Various aspects have to be taken into account:
- minimum and maximum batch sizes for the production of the injection solutions,
- different minimum and maximum down times before bottling,
- set-up sequences,
- restrictions on sterilization,
- to check approved containers or lines,
- interdependencies of the lines,
- and dealing with batch residues, etc.
In addition, there are also special forms of production, such as continuous bottling, parallel preparations or two-stage packaging.
The ORSOFT Manufacturing Workbench has additional modules with which this complex network of restrictions can be mapped and thus an automated campaign formation can be carried out. This reduces the risk of errors when creating campaigns, reduces the effort required for planning, formalizes knowledge and, as a result, allows a wider circle of employees to carry out the demanding planning work.
Contract Manufacturing
Many companies include production for third parties in their business model. The high level of client integration influences the production planning process through provided materials, necessary production shares, client order stocks or frequent adjustments to the process.
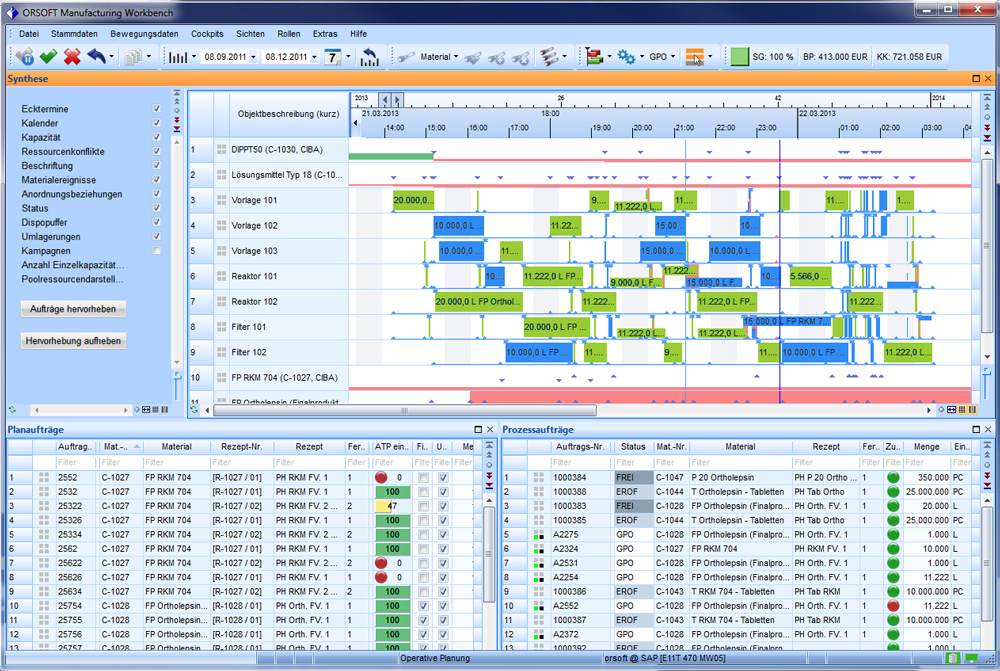
Pharma Synthesis
The processes are characterized by many synthesis steps that build up on one another and are dependent on one another. We have a lot of experience in imaging technical specifics such as fixed ‘paths through the system’ and ‘logical relationship’ for many customers. This also includes campaign handling including setup and cleaning logics.
Batch-based management of logistics processes is a standard within the pharmaceutical industry sector. Functions of ORSOFT Manufacturing Workbench are based on the entire logistic production model, which is fully used, including batch-related information at the levels of stock, production order or batch object.

Shelf Life
Pharmaceutical processes are driven by the temporary applicability of materials and products, which is especially relevant for bio-technological materials and the development of new product characteristics. Reporting and logistical requirements take the duration settings into consideration. As a result, batches close to their expiration date can be identified early in the scheduling process.

Pharmaceutical Packaging
In addition to the manufacturing steps in the areas of bulk production, filling, and synthesis, packaging often turns out to be a planning bottleneck. Unlike the above-mentioned production areas, this last stage typically requires discrete manufacturing techniques in order to identify conflicts at an early phase on multi-purpose packaging machines, to optimally plan orders, and to avoid long-term conflicts.
This ultimately ensures on-time delivery to the customer and is a major contribution to the company’s success.
Typical challenges are alternative facilities, bottlenecks in the availability of personnel, and the provision of appropriate packaging materials. These issues are solved in the standard ORSOFT Manufacturing Workbench detailed planning solution.
